Human Medications, Human Drugs, Animal Medications, Animal Drugs, Pharmacy law, Pharmaceutical law, Compounding law, Sterile and Non Sterile Compounding 797 Compliance, Veterinary law, Veterinary Compounding Law; Health Care; Awareness of all Types of Compounding Issues; Pharmacy Benefit Managers (PBMs), Outsourcing Facilities Food and Drug Administration and Compliance Issues
Saturday, April 10, 2021
Tennesseean sentenced in insurance kickback scheme
Tuesday, April 6, 2021
| 04/06/2021 | 03/26/2021 | RPK Pharmaceuticals Inc. | Center for Drug Evaluation and Research | Failure to Comply with Drug Listing Requirements/Misbranded | ||
| 04/06/2021 | 03/29/2021 | IMAD International, LLC | Division of Northeast Imports | Foreign Supplier Verification Program (FSVP) | ||
| 04/06/2021 | 03/31/2021 | Neoingenium Labs, SA de CV | Center for Drug Evaluation and Research | Finished Pharmaceuticals/Unapproved New Drug/Misbranded/Adulterated | ||
| 04/05/2021 | 04/05/2021 | Van Howling Enterprise LLC d/b/a Driftwood Vapor | Center for Tobacco Products | Family Smoking Prevention and Tobacco Control Act/Adulterated/Misbranded | ||
| 04/05/2021 | 04/05/2021 | Super Vape'z | Center for Tobacco Products | Family Smoking Prevention and Tobacco Control Act/Adulterated/Misbranded | ||
| 04/02/2021 | 04/01/2021 | Natural Adventure, LLC | Center for Drug Evaluation and Research | Unapproved and Misbranded Products Related to Coronavirus Disease 2019 (COVID-19) | ||
| 04/01/2021 | 03/26/2021 | Kealani Distribution LLC/Violet Vapor LLC | Center for Tobacco Products | Family Smoking Prevention and Tobacco Control Act/Adulterated/Misbranded | ||
| 04/01/2021 | 03/26/2021 | Voltage Vapor Shop LLC | Center for Tobacco Products | Family Smoking Prevention and Tobacco Control Act/Adulterated/Misbranded | ||
| 04/01/2021 | 03/26/2021 | Vapors Gold LLC | Center for Tobacco Products | Family Smoking Prevention and Tobacco Control Act/Adulterated/Misbranded |
Friday, April 2, 2021
| 03/30/2021 | 03/26/2021 | Morin Enterprises Inc. d/b/a Ecig Crib | Center for Tobacco Products | Family Smoking Prevention and Tobacco Control Act/Adulterated/Misbranded | ||
| 03/30/2021 | 03/26/2021 | Nicoticket LLC | Center for Tobacco Products | Family Smoking Prevention and Tobacco Control Act/Adulterated/Misbranded | ||
| 03/30/2021 | 03/26/2021 | Grand Vapors | Center for Tobacco Products | Family Smoking Prevention and Tobacco Control Act/Adulterated/Misbranded | ||
| 03/30/2021 | 03/26/2021 | Bouji Moj Oyeeb, Inc. d/b/a MasterMix E-Liquid | Center for Tobacco Products | Family Smoking Prevention and Tobacco Control Act/Adulterated/Misbranded | ||
| 03/30/2021 | 03/26/2021 | Planet Vapor | Center for Tobacco Products | Family Smoking Prevention and Tobacco Control Act/Adulterated/Misbranded | ||
| 03/30/2021 | 03/26/2021 | RNJ Field Inc d/b/a Thunderhead Corporation Fife | Center for Tobacco Products | Family Smoking Prevention and Tobacco Control Act/Adulterated/Misbranded | ||
| 03/30/2021 | 03/30/2021 | The Juice Bar | Center for Tobacco Products | Family Smoking Prevention and Tobacco Control Act/Adulterated/Misbranded | ||
| 03/25/2021 | 03/22/2021 | Realistic Wireless Inc d/b/a Vintage Vapors | Center for Tobacco Products | Family Smoking Prevention and Tobacco Control Act/Adulterated/Misbranded |
| Posted Date | Letter Issue Date | Company Name | Issuing Office | Subject | Response Letter | Closeout Letter |
|---|---|---|---|---|---|---|
| 04/01/2021 | 03/26/2021 | Kealani Distribution LLC/Violet Vapor LLC | Center for Tobacco Products | Family Smoking Prevention and Tobacco Control Act/Adulterated/Misbranded | ||
| 04/01/2021 | 03/26/2021 | Voltage Vapor Shop LLC | Center for Tobacco Products | Family Smoking Prevention and Tobacco Control Act/Adulterated/Misbranded | ||
| 04/01/2021 | 03/26/2021 | Vapors Gold LLC | Center for Tobacco Products | Family Smoking Prevention and Tobacco Control Act/Adulterated/Misbranded | ||
| 03/30/2021 | 11/09/2020 | Tarmac Products, Inc. | Division of Medical Device and Radiological Health Operations Central | CGMP/QSR/Medical Devices/Adulterated | ||
| 03/30/2021 | 03/10/2021 | Trippo International LLC | Center for Food Safety and Applied Nutrition | Dietary Supplement/Adulterated | ||
| 03/30/2021 | 03/16/2021 | Duoc Thao Tre Xanh, LLC | Division of Human and Animal Food Operations East IV | New Drug/Misbranded | ||
| 03/30/2021 | 03/16/2021 | Anna Health, LLC | Division of Human and Animal Food Operations East IV | New Drug/Misbranded | ||
| 03/30/2021 | 03/19/2021 | Lipotriad LLC | Division of Human and Animal Food Operations East II | New Drug/Misbranded | ||
| 03/30/2021 | 03/23/2021 | Distribuidora Lagunera del Norte S.A. de C.V. | Center for Drug Evaluation and Research | Finished Pharmaceuticals/Unapproved New Drug/Misbranded/Adulterated | ||
| 03/30/2021 | 03/26/2021 | Kidney Puncher LLC | Center for Tobacco Products | Family Smoking Prevention and Tobacco Control Act/Adulterated/Misbranded |
Friday, March 26, 2021
Thursday, March 25, 2021
Memorandum of Understanding Addressing Certain Distributions of Compounded Drugs
FDA announced in October 2020 the standard Memorandum of Understanding Addressing Certain Distributions of Compounded Human Drug Products became available for signature by the states. This MOU is an agreement between state boards of pharmacy or other state agencies and FDA. The MOU addresses interstate distribution of inordinate amounts of compounded drugs and complaint investigation by a state regulator relating to compounded drugs distributed outside the state.
FDA developed the MOU in consultation with the National Association of Boards of Pharmacy (NABP), as described in section 503A of the Federal Food, Drug, and Cosmetic Act (FD&C Act). The MOU is a key public health protection in the law and is anticipated to help enhance communication and maximize federal and state resources for oversight of compounded drugs produced by traditional pharmacies.
Reducing the risks associated with compounded drugs
States, along with FDA, play a vital role in helping to reduce the risks associated with compounded drugs, while preserving appropriate patient access. For example, if a compounder distributes compounded drugs to multiple states, it can be difficult to gather information about adverse drug experiences and quality issues associated with those drugs, connect them to the compounder and take coordinated action to address a potentially serious public health problem. Close collaboration among states, and between states and the federal government may help prevent serious and widespread problems by helping to better identify adverse drug experiences and drug quality concerns across the country.
Information sharing network
FDA has entered into a cooperative agreement with NABP to establish an information sharing network available to the states. Through this network, FDA expects that states will have the option to collect, assess and review information about pharmacies in their states and share information with FDA as outlined in the MOU.
This tool is intended to provide an option for states and potentially reduce the resource burden of reporting under the MOU for states that choose to use it.
Expected benefits of the information sharing network:
- Information access: facilitate information sharing between state regulators and FDA on the distribution of compounded drugs interstate and complaints related to drugs compounded in the state and distributed outside the state
- Facilitate collaboration: enhance state and federal oversight and help regulators focus limited resources on compounders that present the greatest risk
- Streamlined identification: enable state regulators to identify pharmacies that distribute an inordinate amount of compounded drugs interstate and report that information to FDA
Complaint investigations
Under the MOU, states agree to investigate complaints about adverse drug experiences and drug quality issues related to drugs compounded at pharmacies within the state and distributed outside the state. States then notify FDA about adverse drug experience and drug quality issues relating to a drug compounded at a pharmacy, if serious, or by a physician as soon as possible, but no later than five business days after the state receives the complaint. This timeframe will facilitate collaboration between states and FDA on serious issues that have the potential to affect patients in multiple states.
Inordinate amounts and the 50% threshold for information sharing
Under the MOU, states agree to share certain information with FDA about pharmacies that distribute more than 50% of their compounded drug prescription orders interstate (see III.b.1 of MOU). The MOU, however, does not place a limit on the distribution of compounded drugs interstate by a pharmacy located in a state that has signed the MOU.
States that sign the MOU also agree to report to FDA if they become aware of a physician who is distributing compounded drugs interstate.
5% statutory limit on distributing compounded drugs out of the state
Section 503A of the FD&C Act limits distribution of compounded drugs outside the state by a licensed pharmacist, pharmacy or physician located in a state that has not signed the MOU to 5% of its total prescription orders dispensed or distributed.
Timeframe for signature
States may sign the MOU at any time. FDA is providing a period of one year, which concludes on October 27, 2021, for states to consider signing the MOU before it intends to enforce the 5% limit in section 503A of the FD&C Act in states that have not signed the MOU. This timeframe should correspond to a full legislative cycle for most states and allows time for states to modify their laws and regulations, if necessary.
Products not covered by the MOU
Products not covered by the MOU include:
- drugs intended for veterinary use
- repackaged drug products
- radiopharmaceuticals
- biological products subject to licensure under section 351 of the Public Health Service Act
- drugs compounded by outsourcing facilities under section 503B of the FD&C Act
Questions
See Memorandum of Understanding Addressing Certain Distributions of Compounded Drugs Questions and Answers for more information. Please email questions regarding the MOU to compounding@fda.hhs.gov.
Additional information
Purpose of Blog
Search This Blog
Featured Post
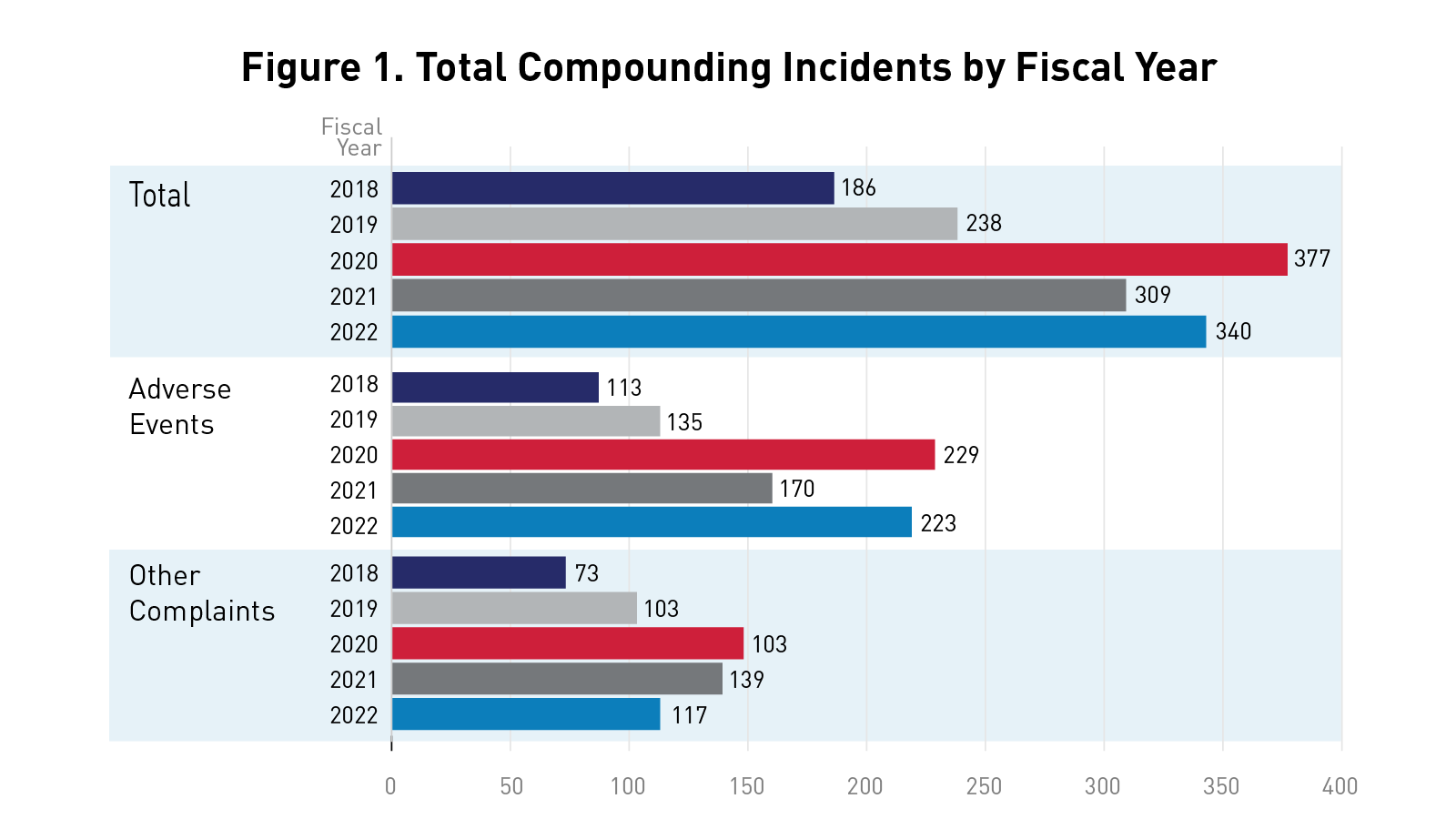
Essential Information: FDA Mitigating Risks of Compounded Drugs Through Surveillance
Mitigating Risks of Compounded Drugs Through Surveillance FDA’s Compounding Incidents Program aims to help protect the public against poor...
|
|